MANUFACTURING
You are here:
- Home
- MANUFACTURING
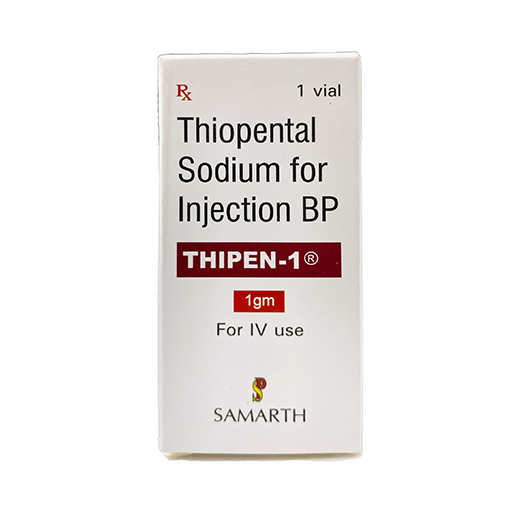
PRODUCT NAME:THIOPENTONE SODIUM INJECTION
BRAND NAME: THIPEN-1000
STRENGTH: 1000 MG
PACKING: SINGLE VIAL PER BOX
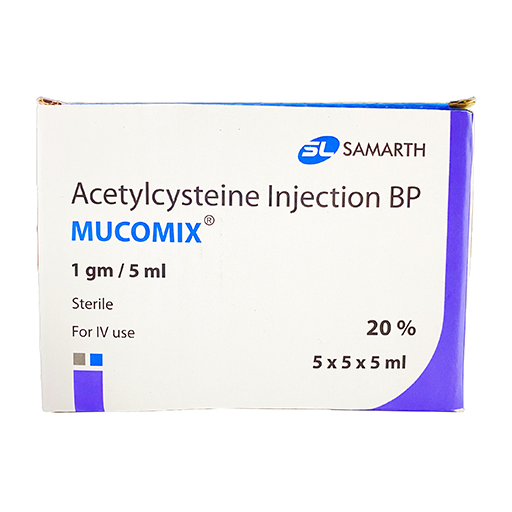
PRODUCT NAME: N- ACETYLCYSTEINE INJECTION
BRAND NAME: MUCOMIX
STRENGTH: 1 GM / 5 ML (20%)
PACKING: 25 AMPOULES PER BOX
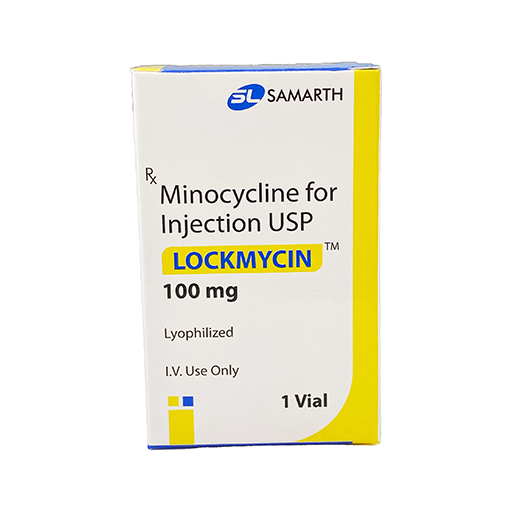
PRODUCT NAME: MINOCYCLINE FOR INJECTION
BRAND NAME: LOCKMYCIN
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: HYALURONIDASE FOR INJECTION (LYOPHILIZED)
BRAND NAME: HINEX
STRENGTH: 1500 IU / 5 ML
PACKING: 10 VIALS PER BOX

PRODUCT NAME: BUPIVACAINE INJECTION
BRAND NAME: NUMBCAINE / BUPIBLOC
STRENGTH: 0.5%, 20 ML
PACKING: SINGLE VIAL PER BOX
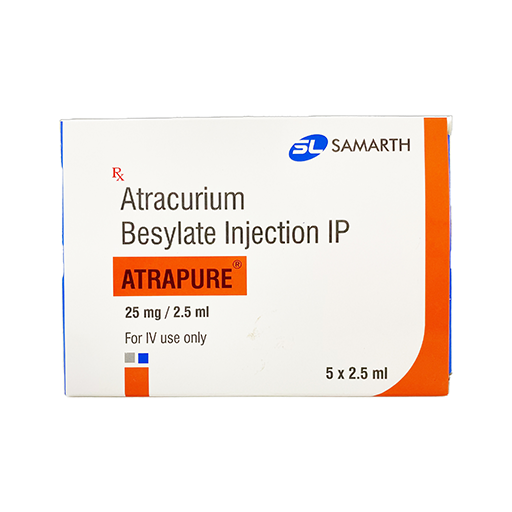
PRODUCT NAME:ATRACURIUM BESYLATE INJECTION
BRAND NAME: ATRAPURE
STRENGTH: 25 MG / 2.5 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: NIFEDIPINE (0.3% W/W) & LIDOCAINE (1.5% W/W) CREAM
BRAND NAME: ANOBLISS
STRENGTH: 30 GM
PACKING: SINGLE TUBE PER BOX
PRODUCT NAME: ISOPRENALINE HCL INJECTION
BRAND NAME: ISOLIN
STRENGTH: 0.2 MG / 1 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME:L-ORNITHINE-L- ASPARTATE INFUSION CONCENTRATE
BRAND NAME: SAMLOL
STRENGTH: 5 GM / 10 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: BENZTROPINE MESYLATE INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 2 MG / 2 ML
PACKING: 5 AMPOULES PER BOX
PRODUCT NAME: CARBOPROST TROMETHAMINE INJECTION
BRAND NAME: CPROSTIN
STRENGTH: 250 MCG / 1 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: AMINOCAPROIC ACID INJECTION
BRAND NAME: HAMOSTAT
STRENGTH: 5 GM / 20 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DOBUTAMINE HCL INJECTION
BRAND NAME: DOBUSTAT
STRENGTH: 250 MG / 20 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PIPERACILLIN SODIUM + TAZOBACTAM SODIUM INJECTION
BRAND NAME: PIROTAZ / PLUGTAZ
STRENGTH: 4 GM + 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PENICILLAMINE CAPSULES
BRAND NAME: PENICITIN
STRENGTH: 250 MG
PACKING: 50 CAPSULES PER BOX

PRODUCT NAME: CEFOPERAZONE SODIUM + SULBACTAM SODIUM INJECTION
BRAND NAME: PURECEF-S
STRENGTH: 1 GM + 0.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: VECURONIUM BROMIDE FOR INJECTION (LYOPHILIZED)
BRAND NAME: SAMVEC-10
STRENGTH: 10 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: UROFOLLITROPIN FOR INJECTION (HIGHLY PURIFIED) (LYOPHILIZED)
BRAND NAME: EUVIFOL-HP
STRENGTH: 150 IU
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: UROFOLLITROPIN FOR INJECTION (HIGHLY PURIFIED) (LYOPHILIZED)
BRAND NAME: EUVIFOL-HP
STRENGTH: 75 IU
PACKING: SINGLE VIAL PER BOX
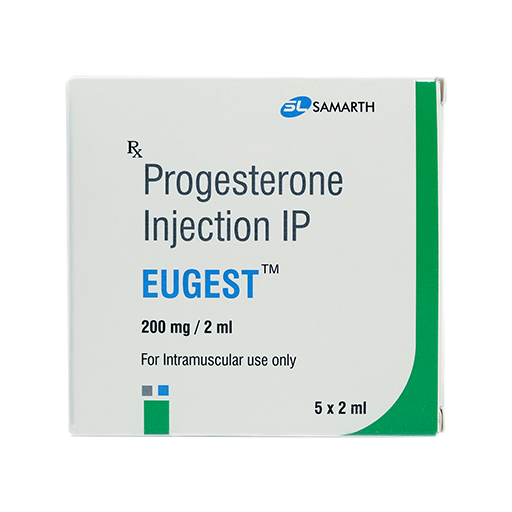
PRODUCT NAME: PROGESTERONE INJECTION
BRAND NAME: EUGEST
STRENGTH: 200 MG / 2 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: MENOTROPIN FOR INJECTION (HIGHLY PURIFIED) (LYOPHILIZED)
BRAND NAME: EUGON-HP
STRENGTH: 150 IU
PACKING: SINGLE VIAL PER BOX
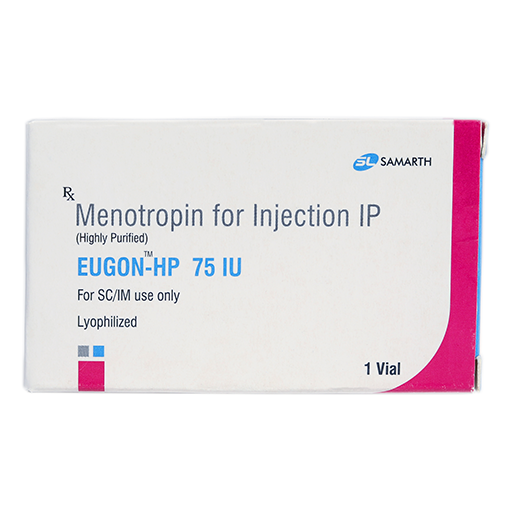
PRODUCT NAME: MENOTROPIN FOR INJECTION (HIGHLY PURIFIED) (LYOPHILIZED)
BRAND NAME: EUGON-HP
STRENGTH: 75 IU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: LEUPROLIDE ACETATE INJECTION
BRAND NAME: EUROLIDE
STRENGTH: 1 MG / 0.5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: LEUPROLIDE ACETATE INJECTION
BRAND NAME: EUROLIDE
STRENGTH: 4 MG / 4 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: LEUPROLIDE ACETATE FOR INJECTION (LYOPHILIZED)
BRAND NAME: LEULIDE
STRENGTH: 11.25 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: LEUPROLIDE ACETATE FOR INJECTION (LYOPHILIZED)
BRAND NAME: EUROLIDE DEPOT / LEULIDE
STRENGTH: 3.75 MG
PACKING: SINGLE VIAL PER BOX
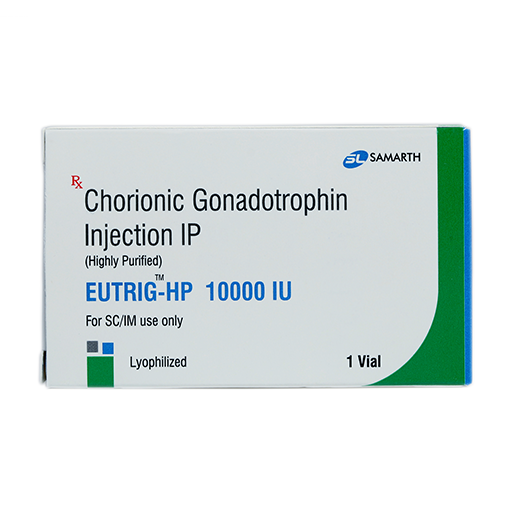
PRODUCT NAME: CHORIONIC GONADOTROPHIN INJECTION (HIGHLY PURIFIED) (LYOPHILIZED)
BRAND NAME: EUTRIG-HP
STRENGTH: 10,000 IU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CHORIONIC GONADOTROPHIN INJECTION (HIGHLY PURIFIED) (LYOPHILIZED)
BRAND NAME: EUTRIG-HP
STRENGTH: 5,000 IU
PACKING: SINGLE VIAL PER BOX
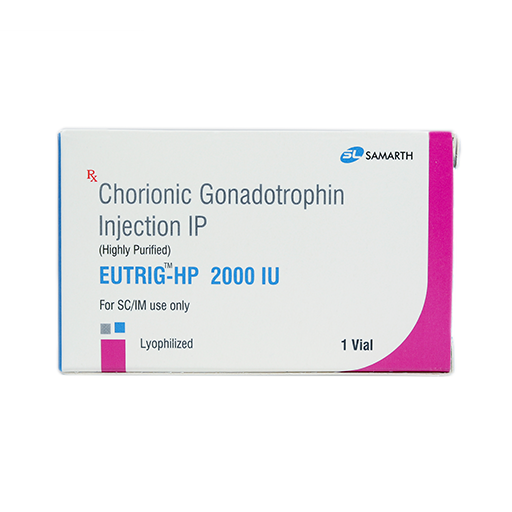
PRODUCT NAME: CHORIONIC GONADOTROPHIN INJECTION (HIGHLY PURIFIED) (LYOPHILIZED)
BRAND NAME: EUTRIG-HP
STRENGTH: 2,000 IU
PACKING: SINGLE PFS PER BOX

PRODUCT NAME: CETRORELIX ACETATE FOR INJECTION
BRAND NAME: EURELIX PFS
STRENGTH: 0.25 MG / 0.5ML
PACKING: SINGLE PFS PER BOX
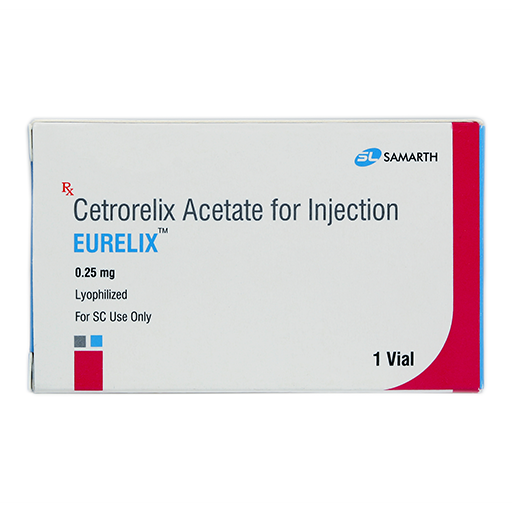
PRODUCT NAME: CETRORELIX ACETATE FOR INJECTION (LYOPHILIZED)
BRAND NAME: EURELIX
STRENGTH: 0.25 MG
PACKING: SINGLE VIAL PER BOX
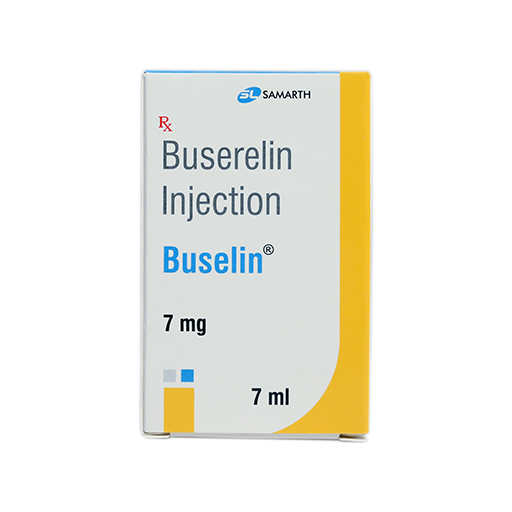
PRODUCT NAME: BUSERELIN ACETATE INJECTION
BRAND NAME: BUSELIN
STRENGTH: 7 MG / 7 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: BUSERELIN ACETATE INJECTION
BRAND NAME: BUSELIN
STRENGTH: 0.5 MG / 0.5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PROGESTERONE INJECTION
BRAND NAME: EUGEST
STRENGTH: 100 MG / 1 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: PROGESTERONE INJECTION
BRAND NAME: EUGEST
STRENGTH: 100 MG / 2 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: HYDROXYPROGESTERONE CAPROATE INJECTION
BRAND NAME: GENRIC NAME
STRENGTH: 250 MG / 1 ML
PACKING: 10 AMPOULES PER BOX
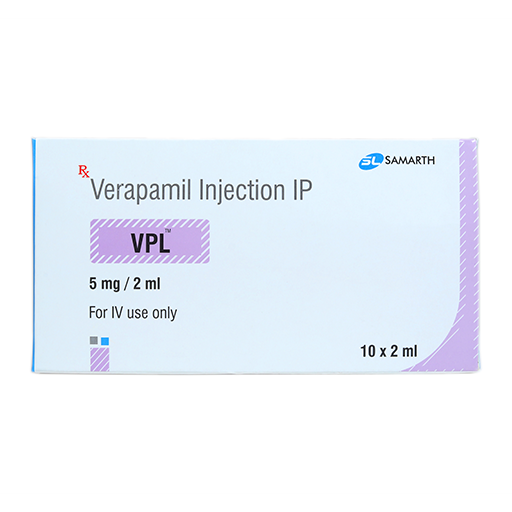
PRODUCT NAME: VERAPAMIL HCL INJECTION
BRAND NAME: VPL
STRENGTH: 5 MG / 2 ML
PACKING: 10 AMPOULES PER BOX
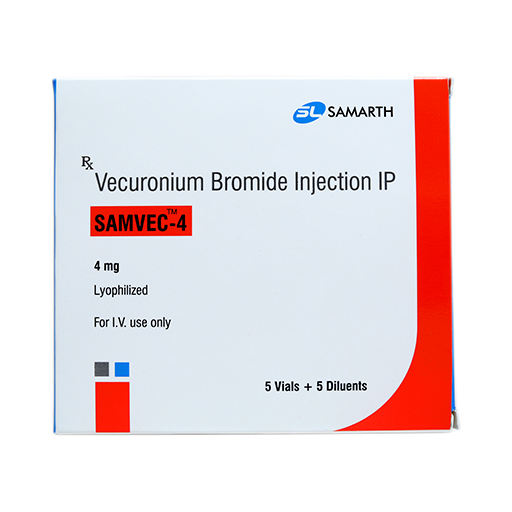
PRODUCT NAME:VECURONIUM BROMIDE FOR INJECTION (LYOPHILIZED)
BRAND NAME: SAMVEC-4
STRENGTH: 4 MG
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: VASOPRESSIN INJECTION (PREFILLED SYRINGE)
BRAND NAME: CPRESSIN
STRENGTH: 40 IU / 1 ML
PACKING: SINGLE PFS PER BOX

PRODUCT NAME: VASOPRESSIN INJECTION
BRAND NAME: CPRESSIN P
STRENGTH: 20 IU / 1 ML
PACKING: 5 AMPOULES PER BOX
PRODUCT NAME: TRANEXAMIC ACID INJECTION
BRAND NAME: TRANEMIC
STRENGTH: 500 MG / 5 ML
PACKING: 25 AMPOULES PER BOX

PRODUCT NAME: STREPTOKINASE FOR INJECTION (LYOPHILIZED)
BRAND NAME: STUKINASE
STRENGTH: 15,00,000 IU
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: SOTALOL HCL TABLETS
BRAND NAME: SOLET
STRENGTH: 40 MG
PACKING: 50 TABLETS PER BOX

PRODUCT NAME: SODIUM NITROPRUSSIDE INJECTION
BRAND NAME: NIPRESS
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ROPIVACAINE HYDROCHLORIDE INJECTION
BRAND NAME: ROPIFAST
STRENGTH:0.75%, 20 ML
PACKING: SINGLE AMPOULE PER BOX
PRODUCT NAME: ROPIVACAINE HYDROCHLORIDE INJECTION
BRAND NAME:ROPIFAST
STRENGTH:0.2%, 20 ML
PACKING: SINGLE AMPOULE PER BOX
PRODUCT NAME: PROTAMINE SULPHATE INJECTION
BRAND NAME: PROTA
STRENGTH:50 MG / 5 ML
PACKING: 5 AMPOULES PER BOX
PRODUCT NAME: PHENYLEPHRINE HCL INJECTION
BRAND NAME: FRENIN
STRENGTH: 10 MG / 1 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: PHENOXYBENZAMINE CAPSULES
BRAND NAME: FENOXENE
STRENGTH: 50 MG / 1 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME:PHENOXYBENZAMINE CAPSULES
BRAND NAME:FENOXENE
STRENGTH: 10 MG
PACKING: 50 CAPSULES PER BOX
PRODUCT NAME:NORADRENALINE BITARTRATE INJECTION
BRAND NAME:ADRENOR
STRENGTH: 4 MG / 2 ML
PACKING: 20 AMPOULES PER BOX

PRODUCT NAME:NITROGLYCERIN INJECTION
BRAND NAME:NITROCIN
STRENGTH: 50 MG /10 ML
PACKING: 5 AMPOULES PER BOX
PRODUCT NAME:NITROGLYCERIN INJECTION
BRAND NAME:NITROCIN
STRENGTH: 25 MG / 5 ML
PACKING: 25 AMPOULES PER BOX

PRODUCT NAME:NALOXONE HCL INJECTION
BRAND NAME: NALOX
STRENGTH: 0.4 MG / 1 ML
PACKING: 5 AMPOULES PER BOX
PRODUCT NAME:MILRINONE LACTATE INJECTION
BRAND NAME: MILICOR
STRENGTH: 10 MG / 10 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME:LEVOSIMENDAN FOR INJECTION (LYOPHILISED)
BRAND NAME: LEVOSIMED
STRENGTH: 12.5 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: LABETALOL HCL INJECTION
BRAND NAME: LOBET
STRENGTH: 20 MG / 4 ML
PACKING: 2 AMPOULES PER BOX
PRODUCT NAME: LABETALOL HCL INJECTION
BRAND NAME: LOBET
STRENGTH: 100 MG / 20 ML
PACKING:SINGLE AMPOULE PER BOX
PRODUCT NAME: ISOPRENALINE HCL INJECTION
BRAND NAME: ISOLIN
STRENGTH: 2 MG / 1 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: IRON SUCROSE INJECTION
BRAND NAME:FEROGEN / ROSEIRON
STRENGTH: 100 MG / 5 ML
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: HEPARIN SODIUM INJECTION
BRAND NAME: CAPRIN
STRENGTH: 25,000 IU / 5 ML
PACKING: 10 VIALS PER BOX

PRODUCT NAME: HEPARIN SODIUM INJECTION
BRAND NAME: CAPRIN
STRENGTH: 5,000 IU / 5 ML
PACKING: 10 VIALS PER BOX

PRODUCT NAME:GLYCOPYRROLATE INJECTION
BRAND NAME: LICOLATE
STRENGTH: 0.2 MG / 1 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: FUROSEMIDE ORAL SOLUTION
BRAND NAME: FUROPED
STRENGTH: 300 MG / 30 ML
PACKING: SINGLE BOTTLE PER BOX
PRODUCT NAME: ESMOLOL HYDROCHLORIDE INJECTION
BRAND NAME: ESOCARD
STRENGTH: 100 MG / 10 ML
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME:ENOXAPARIN SODIUM INJECTION (PREFILLED SYRINGE)
BRAND NAME: LOWCAP
STRENGTH: 60 MG / 0.6 ML
PACKING: SINGLE PFS PER BOX
PRODUCT NAME: ENALAPRIL MALEATE TABLETS
BRAND NAME: ENAPED
STRENGTH: 2.5 MG
PACKING: 150 TABLETS PER BOX
PRODUCT NAME:DOPAMINE HCL INJECTION
BRAND NAME: DOPAR
STRENGTH: 200 MG / 5 ML
PACKING: 25 AMPOULES PER BOX

PRODUCT NAME: DIGOXIN TABLETS
BRAND NAME: DIXIN
STRENGTH: 0.25 MG
PACKING: 200 TABLETS PER BOX
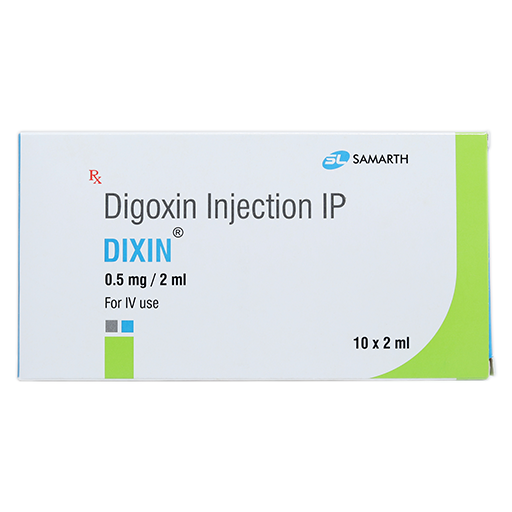
PRODUCT NAME: DIGOXIN INJECTION
BRAND NAME: DIXIN
STRENGTH: 0.5 MG / 2 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: DEXMEDETOMIDINE HYDROCHLORIDE INJECTION
BRAND NAME: DEXMEDINE
STRENGTH: 200 MCG / 2 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: DEXMEDETOMIDINE HYDROCHLORIDE INJECTION
BRAND NAME: DEXMEDINE
STRENGTH: 100 MCG / 1 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: DEXMEDETOMIDINE HYDROCHLORIDE INJECTION
BRAND NAME: DEXMEDINE
STRENGTH: 50 MCG / 0.5 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: CARDIOPLEGIA SOLUTION
BRAND NAME: PLEGIOCARD
STRENGTH: 20 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME:BUPIVACAINE HCL IN DEXTROSE INJECTION
BRAND NAME: NUMBCAINE HEAVY
STRENGTH: 20 MG / 4 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: ATRACURIUM BESYLATE INJECTION
BRAND NAME: ATRAPURE
STRENGTH: 100 MG / 10 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:ATRACURIUM BESYLATE INJECTION
BRAND NAME: ATRAPURE
STRENGTH: 50 MG / 5 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: AMIODARONE TABLETS
BRAND NAME: DURON
STRENGTH: 200 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME:AMIODARONE TABLETS
BRAND NAME: DURON
STRENGTH: 100 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME:AMIODARONE HCL INJECTION
BRAND NAME: DURON
STRENGTH: 150 MG / 3 ML
PACKING: 5 AMPOULES PER BOX
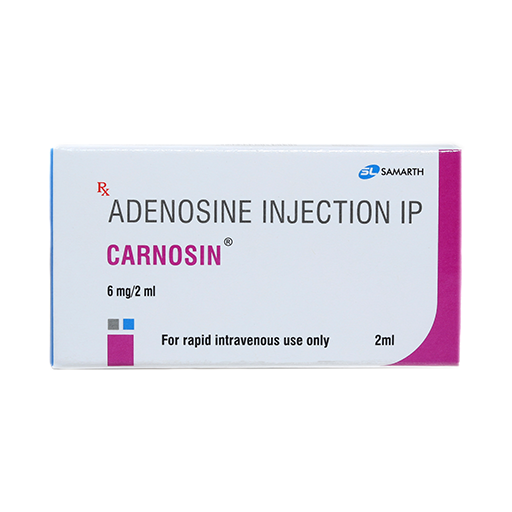
PRODUCT NAME: ADENOSINE INJECTION
BRAND NAME: CARNOSIN
STRENGTH: 6 MG / 2 ML
PACKING: SINGLE AMPOULE PER BOX
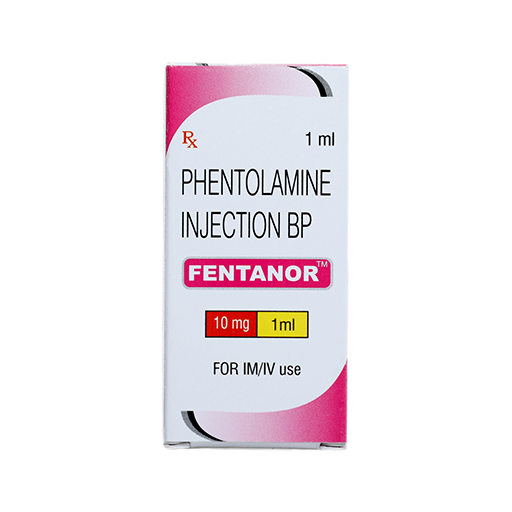
PRODUCT NAME: PHENTOLAMINE MESYLATE INJECTION
BRAND NAME: FENTANOR
STRENGTH: 10 MG / 1 ML
PACKING: SINGLE AMPOULE PER BOX
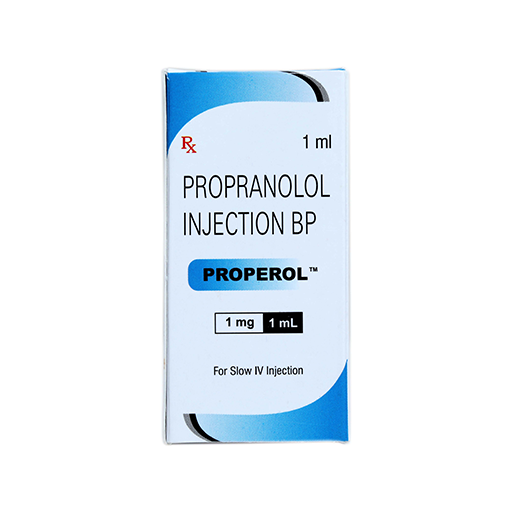
PRODUCT NAME: PROPRANOLOL HCL INJECTION
BRAND NAME: PROPEROL
STRENGTH: 1 MG / 1 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: INDOMETHACIN FOR INJECTION (LYOPHILIZED)
BRAND NAME: DUCTACLOSE
STRENGTH: 1 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: HYDRALAZINE HYDROCHLORIDE INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 20 MG / 1 ML
PACKING: 10 AMPOULES PER BOX
PRODUCT NAME: IMATINIB TABLETS
BRAND NAME: SAMITIB
STRENGTH: 400 MG
PACKING: 30 TABLETS PER BOX
PRODUCT NAME:CYANIDE ANTIDOTE KIT [SODIUM THIOSULPHATE INJECTION 12.5GM/50ML, SODIUM NITRITE INJECTION 300MG/10ML, AMYL NITRITE INHALANT 0.3ML]
BRAND NAME: CYNOCARE
STRENGTH: –
PACKING: KIT
PRODUCT NAME: COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN ACE / COLIMISTA
STRENGTH: 3 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN FORTE / COLIMISTA
STRENGTH: 2 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN / COLIMISTA
STRENGTH: 1 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CLARITHROMYCIN FOR INJECTION (LYOPHILIZED)
BRAND NAME: PLUGCLAR
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CEREBROPROTEIN HYDROLYSATE INJECTION (LYOPHILIZED)
BRAND NAME: CEREBROGEN
STRENGTH: 60 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CEFTAZIDIME INJECTION
BRAND NAME: STEF
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: BETHANECHOL CHLORIDE TABLETS
BRAND NAME: UROTONE DS
STRENGTH: 50 MG
PACKING: 50 TABLETS PER BOX

PRODUCT NAME: BETHANECHOL CHLORIDE TABLETS
BRAND NAME: UROTONE
STRENGTH: 25 MG
PACKING: 50 TABLETS PER BOX

PRODUCT NAME: BACLOFEN TABLETS
BRAND NAME: RICLOFEN
STRENGTH: 10 MG
PACKING: 200 TABLETS PER BOX

PRODUCT NAME: ARTEETHER INJECTION
BRAND NAME: DUNATE
STRENGTH: 60 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ANIDULAFUNGIN INJECTION
BRAND NAME: SAMFUNG
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: AMITRIPTYLINE HCL TABLETS
BRAND NAME: TRIPOXY
STRENGTH: 10 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: ACICLOVIR INTRAVENOUS INFUSION
BRAND NAME: AXOVIR
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ACICLOVIR INTRAVENOUS INFUSION
BRAND NAME: AXOVIR
STRENGTH: 250 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: TRIENTINE HYDROCHLORIDE CAPSULES
BRAND NAME: GENERIC NAME
STRENGTH: 250 MG
PACKING: 100 CAPSULES PER BOX

PRODUCT NAME: PHYTOMENADIONE INJECTION
BRAND NAME: VIT-K1
STRENGTH: 2 MG / 0.2 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: ZOLEDRONIC ACID FOR INJECTION (LYOPHILIZED)
BRAND NAME: ZUNITRA
STRENGTH: 4 MG / 5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: TEMOZOLOMIDE CAPSULES
BRAND NAME: GLIOGEN-250
STRENGTH: 250 MG
PACKING: 5 CAPSULES PER BOX
PRODUCT NAME: SORAFENIB TABLETS
BRAND NAME: SORATIB-200
STRENGTH: 200 MG
PACKING: 30 TABLETS PER BOX / 120 TABLETS PER BOX
PRODUCT NAME: OXALIPLATIN INJECTION
BRAND NAME: OXOPLAN-100
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: METHOTREXATE TABLETS
BRAND NAME: TREX
STRENGTH: 2.5 MG
PACKING: 100 TABLETS PER BOX
PRODUCT NAME: MEGESTROL ACETATE TABLETS
BRAND NAME: ENDACE-160
STRENGTH: 160 MG
PACKING: 30 TABLETS PER BOX

PRODUCT NAME: MEGESTROL ACETATE TABLETS
BRAND NAME: ENDACE-40
STRENGTH: 40 MG
PACKING: 30 TABLETS PER BOX

PRODUCT NAME: LETROZOLE TABLETS
BRAND NAME: LETS / SAMLET / LINEZ
STRENGTH: 2.5 MG
PACKING: 30 TABLETS PER BOX
PRODUCT NAME: LAPATANIB TABLETS
BRAND NAME: ETIBO
STRENGTH: 250 MG
PACKING: 30 TABLETS PER BOX / 150 TABLETS PER BOX

PRODUCT NAME: L-ASPARAGINASE INJECTION
BRAND NAME: L-APARAGIN
STRENGTH: 10,000 IU
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: IMATINIB CAPSULES SAMITIB
BRAND NAME: SAMITIB
STRENGTH: 100 MG
PACKING: 120 CAPSULES PER BOX

PRODUCT NAME: HYDROXYUREA (HYDROXYCARBAMIDE) CAPSULES
BRAND NAME: DUREA
STRENGTH: 500 MG
PACKING: 100 CAPSULES PER BOX

PRODUCT NAME: GEMCITABINE HCL INJECTION
BRAND NAME: LIFOGEM
STRENGTH: 1GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: GEMCITABINE HCL INJECTION
BRAND NAME: LIFOGEM
STRENGTH: 200 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: GEFITINIB TABLETS
BRAND NAME: UNIGEF
STRENGTH: 250 MG
PACKING: 30 TABLETS PER BOX

PRODUCT NAME: FULVESTRANT INJECTION
BRAND NAME: FULVESER
STRENGTH: 250 MG / 5 ML
PACKING: SINGLE PFS PER BOX

PRODUCT NAME: FLUTAMIDE TABLETS
BRAND NAME: FLUTIDE
STRENGTH: 250 MG
PACKING: 30 TABLETS PER BOX

PRODUCT NAME: ENZALUTAMIDE CAPSULES
BRAND NAME: SAMENZA / SAMLUTA
STRENGTH: 40 MG
PACKING: 28 CAPSULES PER BOX / 56 CAPSULES PER BOX

PRODUCT NAME: DOXORUBICIN HCL FOR INJECTION (LYOPHILIZED)
BRAND NAME: SAMRUCIN
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DOCETAXEL INJECTION
BRAND NAME: DOCET
STRENGTH: 120 MG / 3 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DOCETAXEL INJECTION
BRAND NAME: DOCET
STRENGTH: 80 MG / 2 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DOCETAXEL INJECTION
BRAND NAME: DOCET
STRENGTH: 20 MG / 0.5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CALCIUM FOLINATE TABLETS
BRAND NAME: CAPOSTAT
STRENGTH: 500 MG
PACKING: 30 TABLETS PER BOX

PRODUCT NAME: CALCIUM FOLINATE INJECTION
BRAND NAME: CALCIUM LEUCOVORIN
STRENGTH: 15 MG / 2 ML
PACKING: 10 AMPOULES PER

PRODUCT NAME: BICALUTAMIDE TABLETS
BRAND NAME: CALUDEC / SAMTIDE
STRENGTH: 50 MG
PACKING: 30 TABLETS PER BOX / 100 TABLETS PER BOX

PRODUCT NAME: ANASTROZOLE TABLETS
BRAND NAME: ANOSAM
STRENGTH: 1 MG
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: AMPHOTERICIN B FOR INJECTION (LYOPHILIZED)
BRAND NAME: AMPHODEX
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: METHOTREXATE INJECTION
BRAND NAME: TREX
STRENGTH: 50 MG / 2 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: FLUOROURACIL INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 1 GM / 20 ML
PACKING: 5 VIALS PER BOX / SINGLE VIAL PER BOX

PRODUCT NAME: CALCIUM DISODIUM EDETATE INJECTION
BRAND NAME: LEDCURE
STRENGTH: 1 GM / 5 ML
PACKING: 2 AMPOULES PER BOX

PRODUCT NAME: NETILMICIN SULPHATE INJECTION
BRAND NAME: NETICIN
STRENGTH: 300 MG / 3 ML
PACKING: SINGLE AMPOULE PER BOX
PRODUCT NAME: NETILMICIN SULPHATE INJECTION
BRAND NAME: NETICIN
STRENGTH: 200 MG / 2 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: MEROPENEM INJECTION
BRAND NAME: MAXOPEN-500 / EMPIMER
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CYCLOSERINE TABLETS
BRAND NAME: PAMSERINE
STRENGTH: 250 MG
PACKING: 20 TABLETS PER BOX
 PRODUCT NAME:ISOFLURANE LIQUID FOR INHALATION
PRODUCT NAME:ISOFLURANE LIQUID FOR INHALATION
BRAND NAME:IFLURANE-30
STRENGTH: 30 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: NETILMICIN SULPHATE INJECTION
BRAND NAME: NETICIN
STRENGTH: 200 MG / 2 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: NETILMICIN SULPHATE INJECTION
BRAND NAME: NETICIN
STRENGTH: 300 MG / 3 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: MEROPENEM INJECTION
BRAND NAME: MAXOPEN-500 / EMPIMER
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CYCLOSERINE TABLETS
BRAND NAME: PAMSERINE
STRENGTH: 250 MG
PACKING: 20 TABLETS PER BOX

PRODUCT NAME: MICAFUNGIN SODIUM FOR INJECTION (LYOPHILIZED)
BRAND NAME: MYKES
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PHYTOMENADIONE INJECTION
BRAND NAME: KENADION
STRENGTH: 10 MG / 1 ML
PACKING: 25 AMPOULES PER BOX

PRODUCT NAME: BORTEZOMIB FOR INJECTION (LYOPHILIZED)
BRAND NAME: BORTESAM
STRENGTH: 3.5 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: APREPITANT CAPSULES
BRAND NAME: EMECAP
STRENGTH: 125 MG + 80 MG
PACKING: 3 CAPSULES PER BOX

PRODUCT NAME: PHYTOMENADIONE INJECTION
BRAND NAME: KENADION
STRENGTH: 10 MG
PACKING: 20 TABLETS PER BOX
PRODUCT NAME: PHYTOMENADIONE INJECTION
BRAND NAME: KENADION-1
STRENGTH: 1 MG / 0.5 ML
PACKING: 25 AMPOULES PER BOX

PRODUCT NAME: CALCIUM ACETATE TABLETS
BRAND NAME: HYPOPHOS
STRENGTH: 667 MG
PACKING: 100 TABLETS PER BOX
PRODUCT NAME: TEMOZOLOMIDE CAPSULES
BRAND NAME: GLIOGEN-100
STRENGTH: 100 MG
PACKING: 5 CAPSULES PER BOX
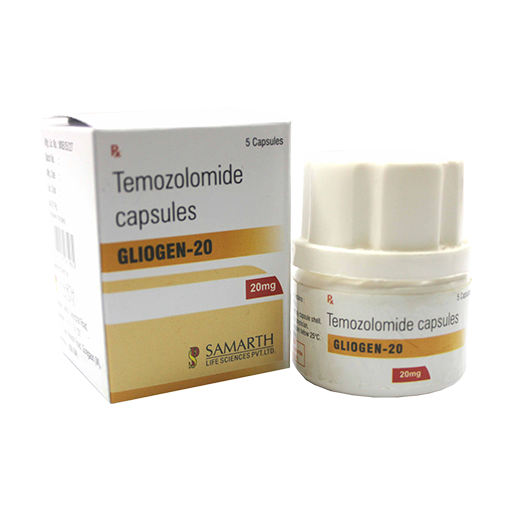
PRODUCT NAME: TEMOZOLOMIDE CAPSULES
BRAND NAME: GLIOGEN-20
STRENGTH: 20 MG
PACKING: 5 CAPSULES PER BOX
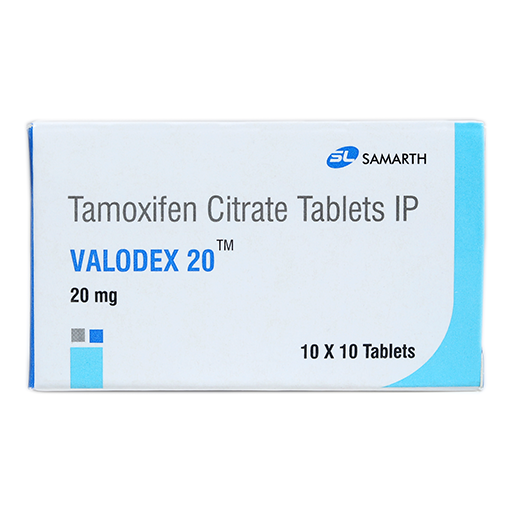
PRODUCT NAME: TAMOXIFEN CITRATE TABLETS
BRAND NAME: VALODEX-20
STRENGTH: 20 MG
PACKING: 100 TABLETS PER BOX
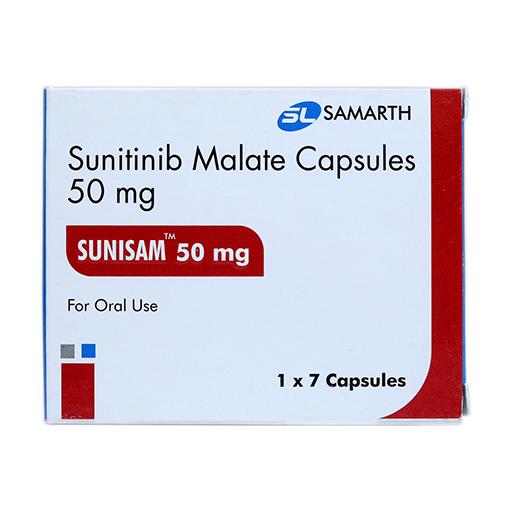
PRODUCT NAME: SUNITINIB MALATE CAPSULES
BRAND NAME: SUNISAM-50
STRENGTH: 50 MG
PACKING: 28 CAPSULES PER BOX

PRODUCT NAME: SUNITINIB MALATE CAPSULES
BRAND NAME: SUNISAM-25
STRENGTH: 25 MG
PACKING: 28 CAPSULES PER BOX

PRODUCT NAME: SUNITINIB MALATE CAPSULES
BRAND NAME: SUNISAM-12.5
STRENGTH: 12.5 MG
PACKING: 28 CAPSULES PER BOX

PRODUCT NAME: PEMETREXED FOR INJECTION
BRAND NAME: PEMAXAM-500
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PEMETREXED FOR INJECTION
BRAND NAME: PEMAXAM-100
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX
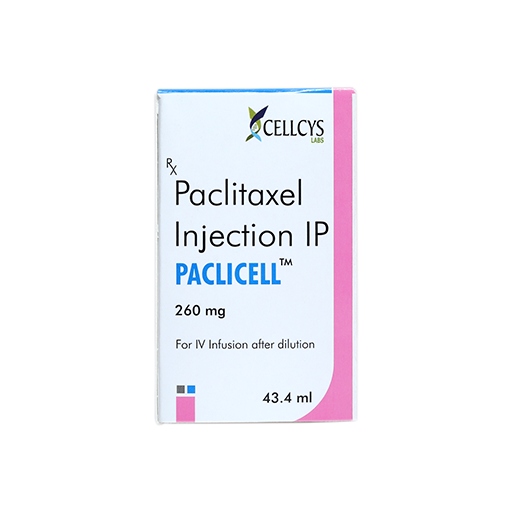
PRODUCT NAME: PACLITAXEL INJECTION
BRAND NAME: PACLICELL
STRENGTH: 260 MG / 43.40 ML
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: PACLITAXEL INJECTION
BRAND NAME: PACLICELL
STRENGTH: 100 MG / 16.67 ML
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: PACLITAXEL INJECTION
BRAND NAME: PACLICELL
STRENGTH: 30 MG / 5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: EXEMESTANE TABLETS
BRAND NAME: SAMEXA
STRENGTH: 25 MG
PACKING: 30 TABLETS PER BOX
PRODUCT NAME: ERLOTINIB TABLETS
BRAND NAME: ERLOSAM
STRENGTH: 150 MG
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: EPIRUBICIN HCL INJECTION (LYOPHILIZED)
BRAND NAME: EPIDOX-10
STRENGTH: 10 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: EPIRUBICIN HCL INJECTION (LYOPHILIZED)
BRAND NAME: EPIDOX-50
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DEXAMETHASONE TABLETS
BRAND NAME: SAMROID-8
STRENGTH: 8 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: DEXAMETHASONE TABLETS
BRAND NAME: SAMROID-4
STRENGTH: 4 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: DASATINIB TABLETS
BRAND NAME: DASATIB-50
STRENGTH: 50 MG
PACKING: 60 TABLETS PER BOX

PRODUCT NAME: DASATINIB TABLETS
BRAND NAME: DASATIB-100
STRENGTH: 100 MG
PACKING: 30 TABLETS PER BOX

PRODUCT NAME: DASATINIB TABLETS
BRAND NAME: DASATIB-70
STRENGTH: 70 MG
PACKING: 60 TABLETS PER BOX
PRODUCT NAME: MERCAPTOPURINE TABLETS
BRAND NAME: MERPURINE
STRENGTH: 50 MG
PACKING: 30 TABLETS PER BOX / 100 TABLETS PER BOX

PRODUCT NAME: MELPHALAN FOR INJECTION (LYOPHILIZED)
BRAND NAME: DUFALAN
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: LEUCOVORIN CALCIUM FOR INJECTION (LYOPHILIZED)
BRAND NAME: GENERIC NAME
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DACARBAZINE FOR INJECTION (LYOPHILIZED)
BRAND NAME: DARZINE
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DACARBAZINE FOR INJECTION (LYOPHILIZED)
BRAND NAME: DARZINE
STRENGTH: 200 MG
PACKING: SINGLE VIAL PER BOX
 PRODUCT NAME: BORTEZOMIB FOR INJECTION (LYOPHILIZED)
PRODUCT NAME: BORTEZOMIB FOR INJECTION (LYOPHILIZED)
BRAND NAME: BORTESAM
STRENGTH: 2 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: SEVOFLURANE LIQUID FOR INHALATION
BRAND NAME: SEVFURANE-50
STRENGTH: 50 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: SEVOFLURANE LIQUID FOR INHALATION
BRAND NAME: SEVFURANE-250
STRENGTH: 250 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: ROCURONIUM BROMIDE INJECTION
BRAND NAME: ROCPURE
STRENGTH: 100 MG / 10 ML
PACKING: SINGLE VIAL PER BOX
 PRODUCT NAME: ROCURONIUM BROMIDE INJECTION
PRODUCT NAME: ROCURONIUM BROMIDE INJECTION
BRAND NAME:ROCPURE
STRENGTH:50 MG / 5 ML
PACKING: SINGLE VIAL PER BOX
 PRODUCT NAME: ISOFLURANE LIQUID FOR INHALATION
PRODUCT NAME: ISOFLURANE LIQUID FOR INHALATION
BRAND NAME: IFLURANE-250
STRENGTH: 250 ML
PACKING: SINGLE BOTTLE PER BOX
 PRODUCT NAME: HEPARIN SODIUM INJECTION
PRODUCT NAME: HEPARIN SODIUM INJECTION
BRAND NAME:CAPRIN FLUSH
STRENGTH: 10 IU / 1 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CISATRACURIUM BESYLATE INJECTION
BRAND NAME: ATRACIS
STRENGTH: 20 MG / 10 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: SUCCINYLCHOLINE CHLORIDE (SUXAMETHONIUM CHLORIDE) FOR INJECTION (LYOPHILIZED)
BRAND NAME: SUXIM
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: VANCOMYCIN HYDROCHLORIDE INJECTION
BRAND NAME: VANTOX-CP
STRENGTH: 1 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: VANCOMYCIN HYDROCHLORIDE INJECTION
BRAND NAME: VANTOX-CP
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: TIGECYCLINE INJECTION
BRAND NAME: TISAM
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: RIFAXIMIN TABLETS
BRAND NAME: RIFORTE
STRENGTH: 550 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: RIFAXIMIN TABLETS
BRAND NAME: RIFORTE
STRENGTH: 400 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: POLYMYXIN B SULPHATE INJECTION
BRAND NAME: POLY-B / POLYIV
STRENGTH: 5,00,000 UNITS
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: PAM (PRALIDOXIME IODIDE) INJECTION
BRAND NAME: ALDOPAM
STRENGTH: 500 MG / 20 ML
PACKING: 2 AMPOULES PER BOX
PRODUCT NAME: IMIPENEM & CILASTATIN INJECTION
BRAND NAME: IMILASTIN / NIMASTIN
STRENGTH: 500 MG + 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN ACE / COLIMISTA
STRENGTH: 3 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN FORTE / COLIMISTA
STRENGTH: 2 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN / COLIMISTA
STRENGTH: 1 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CLARITHROMYCIN FOR INJECTION (LYOPHILIZED)
BRAND NAME: PLUGCLAR
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFTAZIDIME INJECTION
BRAND NAME: STEF
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: FOSFOMYCIN TROMETHAMINE
BRAND NAME: SAMFOS SACHET
STRENGTH: 3 GM
PACKING: 10 SACHETS OF 8 GM PER BOX

PRODUCT NAME: MEROPENEM & SULBACTAM INJECTION
BRAND NAME: PLUGPEN XP / EMPIMER-S / PLUGPEN XP
STRENGTH: 1.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: MEROPENEM INJECTION
BRAND NAME: MAXOPEN-XL / EMPIMER / PLUGPEN
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: NITROFURANTOIN SUSTAINED RELEASE TABLETS
BRAND NAME: NIFCLARE-100
STRENGTH: 100 GM
PACKING: 140 TABLETS PER BOX

PRODUCT NAME: CEFOPERAZONE SODIUM + SULBACTAM SODIUM INJECTION
BRAND NAME: PURECEF-S
STRENGTH: 2 GM + 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFOPERAZONE SODIUM INJECTION
BRAND NAME: PURECEF
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFTAZIDIME & AVIBACTAM POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION
BRAND NAME: STEF AV
STRENGTH: 2.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PIPERACILLIN SODIUM + TAZOBACTAM SODIUM INJECTION
BRAND NAME: PIROTAZ
STRENGTH: 2 GM + 250 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFTRIAXONE + SULBACTAM INJECTION
BRAND NAME: HICEF-S
STRENGTH: 1.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFTRIAXONE + TAZOBACTUM INJECTION
BRAND NAME: HICEF-T
STRENGTH: 1.125 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFTRIAXONE SODIUM INJECTION
BRAND NAME: HICEF
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: POLYMYXIN B SULPHATE INJECTION
BRAND NAME: POLY-B XP
STRENGTH: 7,50,000 UNITS
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN MX / COLIMISTA
STRENGTH: 4.5 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DORIPENEM FOR INJECTION
BRAND NAME: NOZODOR
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ERTAPENEM FOR INJECTION
BRAND NAME: CEDAPENEM
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: FAROPENEM SODIUM EXTENDED-RELEASE TABLETS
BRAND NAME: ULOPEN TABLETS
STRENGTH: 300 MG
PACKING: 6 TABLETS PER BOX

PRODUCT NAME: FOSFOMYCIN SODIUM INJECTION
BRAND NAME: SAMFOS
STRENGTH: 4 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFOPERAZONE SODIUM + SULBACTAM SODIUM INJECTION
BRAND NAME: PURECEF-S
STRENGTH: 0.5 GM + 0.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: BETHANECHOL CHLORIDE SUSTAINEDRELEASE TABLET
BRAND NAME: UROTONE SR
STRENGTH: 75 MG
PACKING: 50 TABLETS PER BOX

PRODUCT NAME: AMOXICILLIN + CLAVULANATE POTASSIUM INJECTION
BRAND NAME: DUCLAV 1.2
STRENGTH: 1 GM + 200 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ERGOCALCIFEROL TABLETS
BRAND NAME: GENERIC NAME
STRENGTH: 10,000 IU (0.25MG)
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: CLOMIFENE TABLETS
BRAND NAME: GENIFENE-50
STRENGTH: 50 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: POLIDOCANOL INJECTION
BRAND NAME: ASKLEROL
STRENGTH: 1%, 3 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: ETHIONAMIDE TABLETS
BRAND NAME: TUMID
STRENGTH: 250 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: CASPOFUNGIN ACETATE FOR INJECTION (LYOPHILIZED)
BRAND NAME: INASTOP-70
STRENGTH: 70 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CALCIUM POLYSTYRENE SULPHONATE POWDER
BRAND NAME: POTBIND
STRENGTH: 300 GM
PACKING: SINGLE JAR

PRODUCT NAME: FLUOROURACIL INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 250 MG / 5 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: AMPHOTERICIN B LIPID COMPLEX INJECTION
BRAND NAME: ANOLIP
STRENGTH: 50MG / 10ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: IBUPROFEN INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 10 MG / 2 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: CETRIMIDE, SODIUM CHLORIDE, POTASSIUM CHLORIDE & CALCIUM CHLORIDE SCRUB SOLUTION, 50 ML
BRAND NAME: LIDAFOAM
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: POLYETHYLENE GLYCOL 400 & PROPYLENE GLYCOL 0.3 % W/V OPHTHALMIC SOLUTION EYE DROPS, 10 ML
BRAND NAME: LATEARS HYDRATE
PACKING: SINGLE BOTTLE PER BOX
PRODUCT NAME: POLYMYXIN B SULFATE, CHLORAMPHENICOL & DEXAMETHASONE SODIUM PHOSPHATE EYE OINTMENT, 5 GM
BRAND NAME: POLYSPORIN DEXA
PACKING: SINGLE TUBE PER BOX

PRODUCT NAME: STREPTOKINASE FOR INJECTION (LYOPHILIZED)
BRAND NAME: STUKINASE
STRENGTH: 7,50,000 IU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: SUCCINYLCHOLINE CHLORIDE (SUXAMETHONIUM CHLORIDE) INJECTION
BRAND NAME: SUXIM
STRENGTH: 100 MG / 2 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: PANCURONIUM INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 4 MG / 2 ML
PACKING: 5 AMPOULES PER BOX
PRODUCT NAME: SUCCINYLCHOLINE CHLORIDE (SUXAMETHONIUM CHLORIDE) INJECTION
BRAND NAME: SUXIM
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME:THIOPENTONE SODIUM INJECTION
BRAND NAME: THIPEN-500
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX
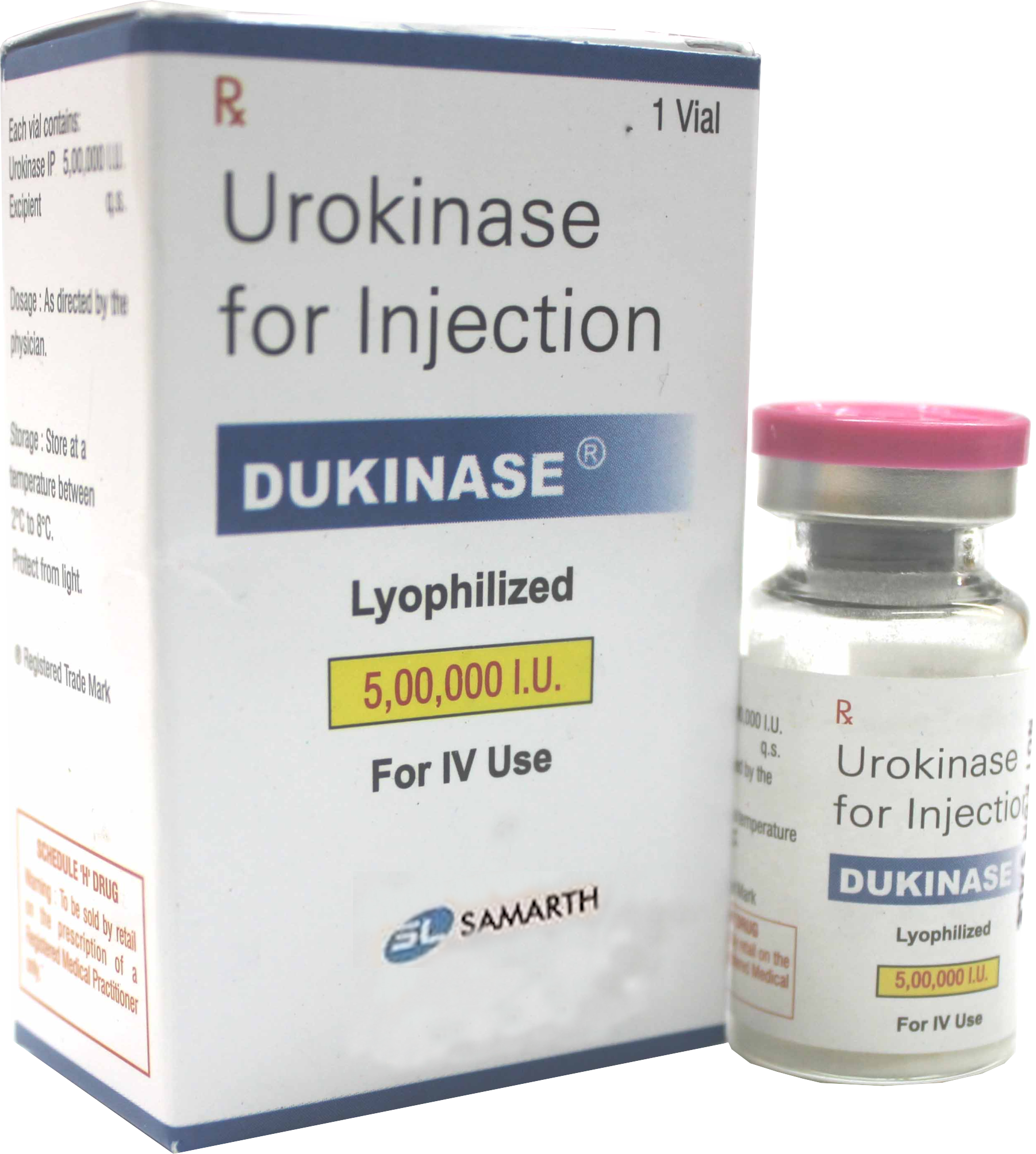
PRODUCT NAME: UROKINASE FOR INJECTION (LYOPHILIZED)
BRAND NAME: DUKINASE
STRENGTH: 5,00,000 IU
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: KETOROLAC TROMETHAMINE INJECTABLE SOLUTION
BRAND NAME: GENERIC NAME
STRENGTH: 30 MG / 1 ML
PACKING: SINGLE VIAL PER BOX
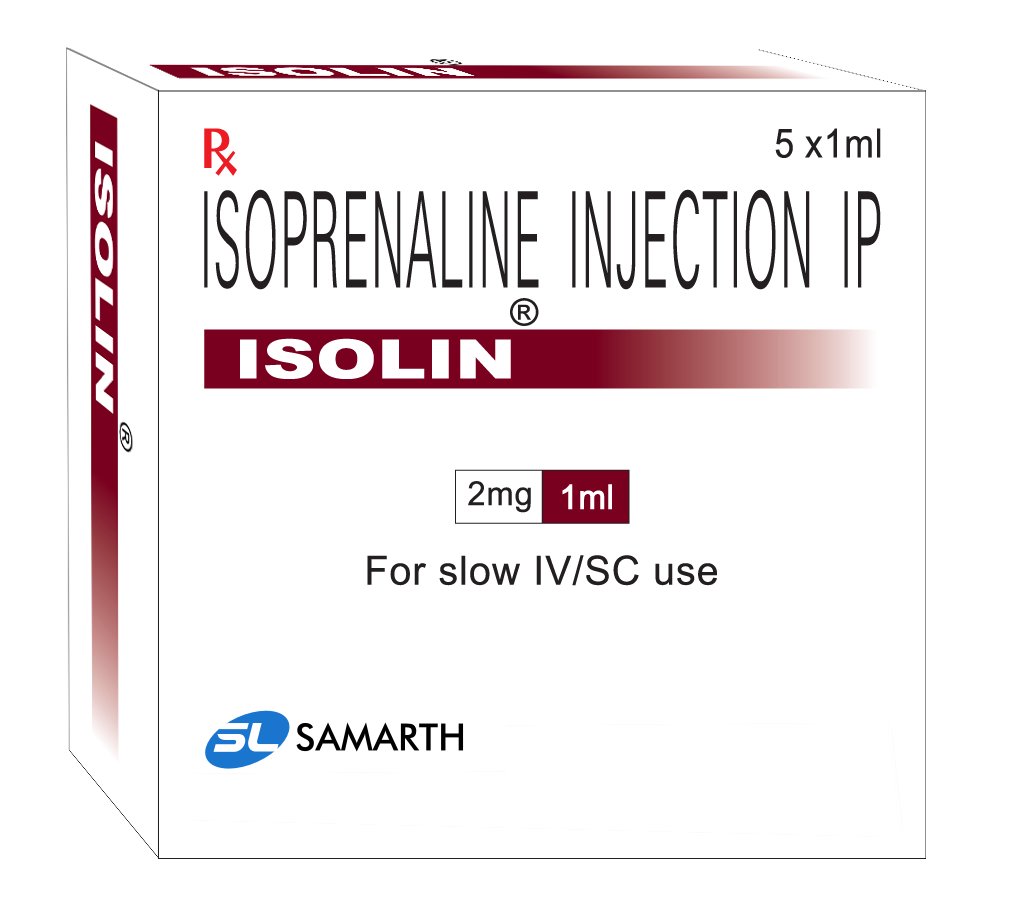
PRODUCT NAME: ISOPRENALINE HCL INJECTION
BRAND NAME: ISOLIN
STRENGTH: 2 MG / 2 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: DIGOXIN TABLETS
BRAND NAME: DIXIN
STRENGTH: 0.125 MG
PACKING: 200 TABLETS PER BOX
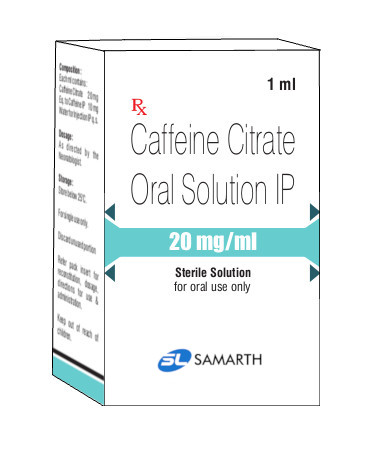
PRODUCT NAME: CAFFEINE CITRATE INJECTION
BRAND NAME: Generic Name
STRENGTH: 20 MG / 1 ML
PACKING: 10 AMPOULES PER BOX
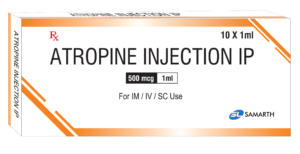
PRODUCT NAME: ATROPINE INJECTION
BRAND NAME: Generic Name
STRENGTH: 500 MCG / 1 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: APROTININ INJECTION (LYOPHILIZED)
BRAND NAME: APROSTAT
STRENGTH: 10,000 KIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: APROTININ INJECTION
BRAND NAME: APROSTAT
STRENGTH: 5,00,00 IU / 50 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CALCIUM FOLINATE TABLETS
BRAND NAME: LEUCORIN
STRENGTH: 15 MG
PACKING: 20 TABLETS PER BOX

PRODUCT NAME: CALCIUM FOLINATE INJECTION
BRAND NAME: CALCIUM LEUCOVORIN
STRENGTH: 50 MG / 5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: SODIUM HYALURONATE EYE DROPS 0.18 % W/V, 10 ML
BRAND NAME: LATEAR FUSION
PACKING: SINGLE BOTTLE PER BOX
PRODUCT NAME: SODIUM HYALURONATE OPHTHALMIC SOLUTION 0.1% W/V, 10 ML
BRAND NAME: LATEARS PF
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: POLYETHYLENE GYCOL 0.4% W/V & PROPYLENE GLYCOL 0.3% W/V EYE DROP, 10 ML
BRAND NAME: POLYVISC
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: NEPAFENAC OPHTHALMIC SUSPENSION 0.1% W/V, 5 ML
BRAND NAME: LIFENEPA
PACKING: SINGLE BOTTLE PER BOX
PRODUCT NAME: MOXIFLOXACIN 0.5% & LOTEPREDNOL ETABONATE OPHTHALMIC 0.5% SUSPENSION, 5 ML
BRAND NAME: MOXIMOXI-LP
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: MOXIFLOXACIN 0.5% & DEXAMETHASONE PHOSPHATE 0.1% EYE DROPS, 5 ML
BRAND NAME: MOXIMOXI-D
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: MOXIFLOXACIN 0.5% EYE DROPS, 5 ML
BRAND NAME: MOXIMOXI
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: POLYMYXIN B SULFATE, CHLORAMPHENICOL & DEXAMETHASONE EYE / EAR DROPS, 5 ML
BRAND NAME: POLYSPORIN DEXA
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: CARBOXYMETHYL CELLULOSE EYE DROPS 1% W/V, 10 ML
BRAND NAME: LACRIVISC DS
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: CARBOXYMETHYL CELLULOSE EYE DROPS, 0.5%, 10 ML
BRAND NAME: LACRIVISC
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: BILBEREY EXTRACT, PINE BARK EXTRACT, LUTEIN, ATAXANTHIN,
ZEAXANTHIN VITAMINS & MINERALS TABLETS
BRAND NAME: ASTALEAF
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: SODIUM HYALURONATE (3% W/V) WITH SODIUM CHONDROITIN SULPHATE (4% W/V)
OPHTHALMIC SOLUTION INJECTION, 1 ML
BRAND NAME: PHACOVISC
PACKING: SINGLE PFS PER BOX
PRODUCT NAME: TAMOXIFEN CITRATE TABLETS
BRAND NAME: VALODEX-10
STRENGTH: 10 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: OXALIPLATIN INJECTION
BRAND NAME: OXOPLAN-50
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: ONDANSETRON INJECTION
BRAND NAME: ONO-4ML
STRENGTH: 8 MG
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: ONDANSETRON INJECTION
BRAND NAME: ONO-2ML
STRENGTH: 4 MG
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: LIPOSOMAL AMPHOTERICIN B FOR INJECTION
BRAND NAME: ANOLIP
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: FILGRASTIM INJECTION (PREFILLED SYRINGE)
BRAND NAME: GRANFILL-300
STRENGTH: 300 MCG / 1 ML
PACKING: SINGLE PFS PER BOX
PRODUCT NAME: ESTRAMUSTINE PHOSPHATE CAPSULES
BRAND NAME: ESTRAMIN
STRENGTH: 140 MG
PACKING: 30 CAPSULE PER BOX

PRODUCT NAME: DOXORUBICIN HCL FOR INJECTION (LYOPHILIZED)
BRAND NAME: SAMRUCIN
STRENGTH: 10 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ABIRATERONE ACETATE TABLETS
BRAND NAME: SAMTICA / ABISAM
STRENGTH: 250 MG
PACKING: 30 TABLETS PER BOX / 120 TABLETS PER BOX

PRODUCT NAME: PROGESTERONE CAPSULES
BRAND NAME: EUGEST-400
STRENGTH: 400 MG
PACKING: 30 CAPSULES PER BOX

PRODUCT NAME: PROGESTERONE CAPSULES
BRAND NAME: EUGEST-200
STRENGTH: 200 MG
PACKING: 30 CAPSULES PER BOX

PRODUCT NAME: PROGESTERONE CAPSULES
BRAND NAME: EUGEST-100
STRENGTH: 100 MG
PACKING: 30 CAPSULES PER BOX
PRODUCT NAME: DIENOGEST TABLETS
BRAND NAME: DEINEST
STRENGTH: 2 MG
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: DEHYDROEPIANDROSTERONE SUSTAINED RELEASE TABLETS
BRAND NAME: OVISERV-SR
STRENGTH: 75 MG
PACKING: 7 TABLETS PER BOX
PRODUCT NAME: LEVOCARNITINE INJECTION
BRAND NAME: VOCARTINE
STRENGTH: 1 GM
PACKING:5 AMPOULES PER BOX
PRODUCT NAME: GLYSERINE 10% W/V & MANNITOL 10 % W/V
BRAND NAME: OSMOPRES
STRENGTH: 100 ML
PACKING: SINGLE BOTTLE PER BOX
PRODUCT NAME: SODIUM BICARBONATE INJECTION
BRAND NAME: SODABICARB
STRENGTH: 8.4% W/V, 25 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: THALIDOMIDE CAPSULES
BRAND NAME: THALOGEN
STRENGTH: 100 MG
PACKING: 30 CAPSULES PER BOX

PRODUCT NAME: THALIDOMIDE CAPSULES
BRAND NAME: THALOGEN
STRENGTH: 50 MG
PACKING: 30 CAPSULES PER BOX

PRODUCT NAME: PACLITAXEL INJECTION
BRAND NAME: DUTAXEL
STRENGTH: 300 MG / 50 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: METHOTREXATE INJECTION
BRAND NAME: TREX
STRENGTH: 500 MG / 20 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: LEUCOVORIN CALCIUM INJECION
BRAND NAME: GENERIC NAME
STRENGTH: 300 MG / 30 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: LEUCOVORIN CALCIUM FOR INJECTION (LYOPHILIZED)
BRAND NAME: GENERIC NAME
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: FLUOROURACIL INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 500 MG / 10 ML
PACKING: 5 VIALS PER BOX / SINGLE VIAL PER BOX

PRODUCT NAME: ETOPOSIDE INJECTION
BRAND NAME: PLACID
STRENGTH: 100 MG / 5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ETOPOSIDE CAPSULES
BRAND NAME: PLACID
STRENGTH: 25 MG
PACKING: 4 CAPSULES PER BOX

PRODUCT NAME: BENDAMUSTINE HYDROCHLORIDE FOR INJECTION (LYOPHILIZED)
BRAND NAME: GENERIC NAME
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: VORICONAZOLE FOR INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 200 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PHENYTOIN INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 250 MG / 5 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: PHENYTOIN INJECTION
BRAND NAME: PHENYTOIN
STRENGTH: 100 MG / 2 ML
PACKING: 100 AMPOULES PER BOX

PRODUCT NAME: FLUORESCEIN SODIUM INJECTION
BRAND NAME: FLURESIN
STRENGTH: 500 MG / 5 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: ETHANOLAMINE OLEATE INJECTION
BRAND NAME: ETHANOLATE
STRENGTH: 5%, 5 ML
PACKING: 2 AMPOULES PER BOX

PRODUCT NAME: ERTAPENEM FOR INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: ERGOCALCIFEROL INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 2,00,000 IU / 1 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: DAPTOMYCIN FOR INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 350 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CHOLESTYRAMINE FOR ORAL SUSPENSION
BRAND NAME: HEPAMIN
STRENGTH: 4 GM
PACKING: SINGLE SACHET

PRODUCT NAME: CASPOFUNGIN ACETATE FOR INJECTION (LYOPHILIZED)
BRAND NAME: INASTOP-50
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CALCIUM POLYSTYRENE SULPHONATE POWDER
BRAND NAME: POTBIND
STRENGTH: 15 GM
PACKING: SINGLE SACHET

PRODUCT NAME: ACYCLOVIR SODIUM TABLETS
BRAND NAME: AXOVIR
STRENGTH: 800 MG
PACKING: 20 TABLETS PER BOX

PRODUCT NAME: ACYCLOVIR SODIUM TABLETS
BRAND NAME: AXOVIR
STRENGTH: 400 MG
PACKING: 20 TABLETS PER BOX

PRODUCT NAME: ACYCLOVIR SODIUM TABLETS
BRAND NAME: AXOVIR
STRENGTH: 200 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: MEDROXYPROGESTERONE ACETATE FOR INJECTABLE SUSPENSION
BRAND NAME: GENRIC NAME
STRENGTH: 150 MG / 1 ML
PACKING: SINGLE PFS PER BOX / SINGLE VIAL PER BOX

PRODUCT NAME: CLOMIFENE TABLETS
BRAND NAME: GENIFENE-100
STRENGTH: 100 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: SPIRONOLACTONE TABLETS
BRAND NAME: ALDOTONE
STRENGTH: 50 MG
PACKING: 150 TABLETS PER BOX

PRODUCT NAME: VITAMIN D3 ORAL SOLUTION
BRAND NAME: SAMSHOT-D3
STRENGTH: 60,000 IU / 5 ML
PACKING: 4 VIALS PER BOX

PRODUCT NAME: VANCOMYCIN HYDROCHLORIDE INJECTION
BRAND NAME: VANTOX-CP
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: VANCOMYCIN HYDROCHLORIDE INJECTION
BRAND NAME: VANTOX-CP
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: URSODEOXYCHOLIC ACID TABLETS
BRAND NAME: UCLICK-300
STRENGTH: 300 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: URSODEOXYCHOLIC ACID TABLETS
BRAND NAME: UCLICK-150
STRENGTH: 150 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: ULIPRISTAL ACETATE TABLETS
BRAND NAME: SAMPRISTAL
STRENGTH: 5 MG
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: ULINASTATIN INJECTION
BRAND NAME: GENSTATIN
STRENGTH: 1,00,000 UNITS
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: TIGECYCLINE INJECTION
BRAND NAME: TISAM
STRENGTH: 50 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: TICARCILLIN & CLAVULANIC ACID FOR INJECTION
BRAND NAME: PLUGFIX
STRENGTH: 3.1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: TERLIPRESSIN INJECTION
BRAND NAME: TERLISTAT
STRENGTH: 1 MG / 10 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: TEICOPLANIN FOR INJECTION (LYOPHILIZED)
BRAND NAME: TEXOPLAN
STRENGTH: 400 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: TAMSULOSIN HCL (MODIFIED RELEASE) & DUTASTERIDE TABLETS
BRAND NAME: DUTALOSIN
STRENGTH: 0.4 MG + 0.5 MG
PACKING: 15 TABLETS PER BOX

PRODUCT NAME: TAMSULOSIN HCL CAPSULES
BRAND NAME:MONOLOSIN-DFT
STRENGTH:0.4 MG + 30 MG
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: TAMSULOSIN HCL CAPSULES
BRAND NAME: MONOLOSIN
STRENGTH: 400 MCG
PACKING: 15 CAPSULES PER BOX

PRODUCT NAME: TADALAFIL TABLETS
BRAND NAME: ALFIL-20
STRENGTH: 20 MG
PACKING: 40 TABLETS PER BOX

PRODUCT NAME: TADALAFIL TABLETS
BRAND NAME: ALFIL-10
STRENGTH: 10 MG
PACKING: 40 TABLETS PER BOX

PRODUCT NAME: TADALAFIL TABLETS
BRAND NAME: ALFIL-5
STRENGTH: 5 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: SUCRALFATE 7%, METRONIDAZOLE 1% & LIGNOCAINE 4% CREAM
BRAND NAME:ANOSUM
STRENGTH: 20 GM
PACKING: SINGLE TUBE PER BOX

PRODUCT NAME: SOLIFENACIN TABLETS
BRAND NAME: SAMLIFEN-10
STRENGTH: 10 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: SOLIFENACIN TABLETS
BRAND NAME: SAMLIFEN-5
STRENGTH: 5 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: SOFOSBUVIR + VELPATASVIR TABLETS
BRAND NAME: VELPAS-S
STRENGTH: 400 MG + 100 MG
PACKING: 28 TABLETS PER BOX

PRODUCT NAME: SODIUM THIOSULPHATE INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 12.5 GM / 50 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: SODIUM TETRADECYL SULPHATE INJECTION
BRAND NAME: SETROL
STRENGTH: 3%, 2 ML
PACKING: 2 AMPOULES PER BOX

PRODUCT NAME: SODIUM NITRITE INJECTION
BRAND NAME: GENERIC NAME
STRENGTH: 300 MG / 10 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: SILODOSIN + DUTASTERIDE TABLETS
BRAND NAME: SILOSAM-D8
STRENGTH: 8 MG + 0.5 MG
PACKING: 150 TABLETS PER BOX

PRODUCT NAME: SILODOSIN + DUTASTERIDE TABLETS
BRAND NAME: SILOSAM-D4
STRENGTH: 4 MG + 0.5 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: SILODOSIN TABLETS
BRAND NAME: SILOSAM-8
STRENGTH: 8 MG
PACKING: 150 TABLETS PER BOX

PRODUCT NAME: SILODOSIN TABLETS
BRAND NAME: SILOSAM-4
STRENGTH: 4 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: RIFAXIMIN TABLETS
BRAND NAME: RIFORTE
STRENGTH: 550 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: RIFAXIMIN TABLETS
BRAND NAME: RIFORTE
STRENGTH: 400 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: RIBAVIRIN CAPSULES
BRAND NAME: RIBAMAX
STRENGTH: 200 MG
PACKING: 40 CAPSULES PER BOX

PRODUCT NAME: PYRIDOSTIGMINE BROMIDE TABLETS
BRAND NAME: DISTINON
STRENGTH: 60 MG
PACKING: 50 TABLETS PER BOX

PRODUCT NAME: PRUCALOPRIDE TABLETS
BRAND NAME: C2WIN
STRENGTH: 2 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: PRUCALOPRIDE TABLETS
BRAND NAME: C2WIN
STRENGTH: 1 MG
PACKING: 100 TABLETS PER BOX

PRODUCT NAME: POTASSIUM CITRATE, MAGNESIUM CITRATE & VIT. B6 ORAL SYRUP
BRAND NAME: LIZERTON
STRENGTH: 450 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: POTASSIUM CITRATE, MAGNESIUM CITRATE & VIT. B6 ORAL SYRUP
BRAND NAME: LIZERTON
STRENGTH: 200 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: POLYMYXIN B SULPHATE INJECTION
BRAND NAME: POLY-B XP
STRENGTH: 7,50,000 UNITS
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: POLYMYXIN B SULPHATE INJECTION
BRAND NAME: POLY-B / POLYIV
STRENGTH: 5,00,000 UNITS
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: POLIDOCANOL INJECTION
BRAND NAME: ASKLEROL
STRENGTH: 3%, 2 ML
PACKING: 2 AMPOULES PER BOX

PRODUCT NAME: PIPERACILLIN SODIUM + TAZOBACTAM SODIUM INJECTION
BRAND NAME: PIROTAZ / PLUGTAZ
STRENGTH: 4 GM + 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PIPERACILLIN SODIUM + TAZOBACTAM SODIUM INJECTION
BRAND NAME: PIROTAZ
STRENGTH: 2 GM + 250 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: PHENOBARBITONE INJECTION
BRAND NAME: FENOBARB
STRENGTH: 200 MG / 1 ML
PACKING: 10 AMPOULES PER BOX

PRODUCT NAME: PENICILLAMINE CAPSULES
BRAND NAME: PENICITIN
STRENGTH: 250 MG
PACKING: 50 CAPSULES PER BOX

PRODUCT NAME: PAM (PRALIDOXIME IODIDE) INJECTION
BRAND NAME: ALDOPAM
STRENGTH: 500 MG / 20 ML
PACKING: 2 AMPOULES PER BOX

PRODUCT NAME: OCTREOTIDE INJECTION
BRAND NAME: ACTIDE
STRENGTH: 100 MCG / 1 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: OCTREOTIDE INJECTION
BRAND NAME: ACTIDE
STRENGTH: 50 MCG / 1 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME: NITROFURANTOIN SUSTAINED RELEASE TABLETS
BRAND NAME: NIFCLARE-100
STRENGTH: 100 MG
PACKING: 140 TABLETS PER BOX

PRODUCT NAME: N-BUTYLCYANOACRYLATE 1ML, SCLEROTHERAPY NEEDLE-21G-1 UNIT,
CLEANSING FLUID-1BOTTLE & SYRINGES-2UNITS
BRAND NAME: ENDOCRYL KIT
STRENGTH: 1 ML KIT
PACKING: KIT

PRODUCT NAME: N-BUTYLCYANOACRYLATE
BRAND NAME: ENDOCRYL
STRENGTH: 1 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: N-BUTYLCYANOACRYLATE
BRAND NAME: ENDOCRYL
STRENGTH: 0.5 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: ACEBROPHYLLINE & ACETYLCYSTEINE TABLETS
BRAND NAME: MUCOMIX-ACE
STRENGTH: 100 MG + 600 MG
PACKING: 20 TABLETS PER BOX

PRODUCT NAME: ACETYLCYSTEINE TABLETS
BRAND NAME: MUCOMIX-ET
STRENGTH: 600 MG
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: ACETYLCYSTEINE TABLETS
BRAND NAME: MUCOMIX
STRENGTH: 600 MG
PACKING: 20 TABLETS PER BOX

PRODUCT NAME: N- ACETYLCYSTEINE INJECTION
BRAND NAME: MUCOMIX
STRENGTH: 400 MG / 2 ML (20%)
PACKING: 20 AMPOULES PER BOX

PRODUCT NAME: N- ACETYLCYSTEINE INJECTION
BRAND NAME: MUCOMIX
STRENGTH: 200 MG / 1 ML (20%)
PACKING: 25 AMPOULES PER BOX

PRODUCT NAME:MYOINOSITOL, D-CHIRO-INOSITOL, VITAMIN D3 & CHROMIUM PICOLINATE TABLETS
BRAND NAME: SALUDOVA
STRENGTH: 550 MG + 13.8 MG + 200 IU + 100 MCG
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: METHYLPREDNISOLONE SODIUM SUCCINATE FOR INJECTION
BRAND NAME: MYPRED
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: METHYLPREDNISOLONE SODIUM SUCCINATE FOR INJECTION
BRAND NAME: MYPRED
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: METHYLPREDNISOLONE SODIUM SUCCINATE FOR INJECTION
BRAND NAME: MYPRED
STRENGTH: 125 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: METHYLPREDNISOLONE SODIUM SUCCINATE FOR INJECTION
BRAND NAME: MYPRED
STRENGTH: 40 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: METHYLCOBALAMIN 750MG, ADENOSYLCOBALAMIN 750MG,
VITAMINS B1,B2,B6 & FOLIC ACID 1.5MG CAPSULES
BRAND NAME: KEY B12
STRENGTH: 750 MG + 750 MG + 5 MG +5 MG + 3 MG + 1.5 MG
PACKING: 15 CAPSULES PER BOX

PRODUCT NAME: METHYLENE BLUE INJECTION
BRAND NAME: SAM-MB
STRENGTH: 100 MG / 10 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME:MEROPENEM & SULBACTAM INJECTION
BRAND NAME: PLUGPEN XP / EMPIMER-S / PLUGPEN XP
STRENGTH: 1.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: MEROPENEM INJECTION
BRAND NAME: MAXOPEN-XL / EMPIMER / PLUGPEN
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: L-ORNITHINE-L- ASPARTATE SACHET
BRAND NAME: SAMLOL
STRENGTH: 5 GM
PACKING: 10 SACHETS

PRODUCT NAME:L-CARNITINE, L-TARTRATE,COENZYME Q10, ZINC SULPHATE
MONOHYDRATE,
LYCOPENE & CUCUMIS MELO EXTRACT TABLETS
BRAND NAME: CPR-M
STRENGTH: –
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: LACTITOL MONOHYDRATE AND ISPAGHULA HUSK GRANULES
BRAND NAME: SOFOLYTE / SAMFIBRE
STRENGTH: 180 MG
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: LACTITOL MONOHYDRATE AND ISPAGHULA HUSK GRANULES
BRAND NAME: SOFOLYTE / SAMFIBRE
STRENGTH: 90 MG
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME:LACTIC ACID WITH SEA BUCKTHORN OIL,TEA TREE OIL & CRANBERRY
BRAND NAME: CLIMATIN
STRENGTH: 100 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME:IMIPENEM & CILASTATIN INJECTION
BRAND NAME: IMILASTIN / NIMASTIN
STRENGTH: 500 MG + 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: HYDROCORTISONE SODIUM SUCCINATE INJECTION
BRAND NAME: HISONE
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: HYDROCORTISONE TABLETS
BRAND NAME: HISONE
STRENGTH: 20 MG
PACKING: 50 TABLETS PER BOX

PRODUCT NAME: HYDROCORTISONE TABLETS
BRAND NAME: HISONE
STRENGTH: 10 MG
PACKING: 200 TABLETS PER BOX

PRODUCT NAME: HYDROCORTISONE TABLETS
BRAND NAME: HISONE
STRENGTH: 5 MG
PACKING: 200 TABLETS PER BOX

PRODUCT NAME:HUMAN NORMAL IMMUNOGLOBULIN
BRAND NAME: HUMANIG
STRENGTH: 5%, 100 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: HUMAN NORMAL ALBUMIN
BRAND NAME: PLASVEX
STRENGTH: 20%, 100 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: HUMAN HEPATITIS B IMMUNOGLOBULIN INJECTION SOLUTION
BRAND NAME: SAMHEP
STRENGTH: 100 IU / 1 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: GLUTATHIONE FOR INJECTION (LYOPHILIZED)
BRAND NAME: GTLIVE
STRENGTH: 600 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: FOSFOMYCIN SODIUM INJECTION
BRAND NAME: SAMFOS
STRENGTH: 4 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: FLUORESCEIN SODIUM INJECTION
BRAND NAME: FLURESIN
STRENGTH: 600 MG / 3 ML
PACKING: 5 AMPOULES PER BOX

PRODUCT NAME: FLUDROCORTISONE ACETATE TABLETS
BRAND NAME: FLORICOT
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:FERROUS ASCORBATE (SR), L-METHYL FOLATE, MECOBALAMIN, BIOTIN, CHOLECALCIFEROL, PYRIDOXAL- 5-PHOSPHATE, ZINC & 8 MINERALS
BRAND NAME: ELREDIN GRANULES
STRENGTH: –
PACKING: 20 SACHETS

PRODUCT NAME:FERROUS ASCORBATE (SR), L-METHYL FOLATE, MECOBALAMIN, BIOTIN, CHOLECALCIFEROL, PYRIDOXAL- 5-PHOSPHATE, ZINC & 8 MINERALS
BRAND NAME: ELREDIN
STRENGTH: 100 MG
PACKING: 15 TABLETS PER BOX

PRODUCT NAME: DOXYCYCLINE INJECTION
BRAND NAME: PLUGDOX
STRENGTH: 100 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:DORIPENEM FOR INJECTION
BRAND NAME: NOZODOR
STRENGTH: 500 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DIMERCAPROL INJECTION
BRAND NAME: BAL
STRENGTH: 100 MG / 2 ML
PACKING: 2 AMPOULES PER BOX

PRODUCT NAME:COLISTIMETHATE SODIUM INJECTION
BRAND NAME: ELISTIN MX / COLIMISTA
STRENGTH: 4.5 MIU
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFTRIAXONE + TAZOBACTUM INJECTION
BRAND NAME: HICEF-T
STRENGTH: 1.125 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CEFTRIAXONE + SULBACTAM INJECTION
BRAND NAME: HICEF-S
STRENGTH: 1.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CEFTRIAXONE SODIUM INJECTION
BRAND NAME: HICEF
STRENGTH: 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFOPERAZONE SODIUM INJECTION
BRAND NAME: PURECEF-S
STRENGTH: 2 GM + 1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFOPERAZONE SODIUM INJECTION
BRAND NAME: PURECEF-S
STRENGTH: 1 GM + 0.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: CEFOPERAZONE SODIUM INJECTION
BRAND NAME: PURECEF-S
STRENGTH:0.5 GM + 0.5 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CEFOPERAZONE SODIUM INJECTION
BRAND NAME: PURECEF
STRENGTH:1 GM
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CALCIUM CARBONATE (CORAL FORM) & VITAMIN D3 TABLETS
BRAND NAME: OSIROC
STRENGTH:500 MG + 500 IU
PACKING: 10 TABLETS PER BOX

PRODUCT NAME: ARTEETHER INJECTION
BRAND NAME: DUTHER
STRENGTH: 150 MG / 2 ML
PACKING: 3 AMPOULES PER BOX

PRODUCT NAME: AMYL NITRITE INHALANT
BRAND NAME: GENERIC NAME
STRENGTH: 0.3 ML
PACKING: SINGLE AMPOULE PER BOX

PRODUCT NAME:AMOXICILLIN + CLAVULANATE POTASSIUM INJECTION
BRAND NAME: DUCLAV 1.2
STRENGTH: 1 GM + 200 MG
PACKING: SINGLE VIAL PER BOX
PRODUCT NAME: DEHYDROEPIANDROSTERONE CAPSULES
BRAND NAME: OVISERV
STRENGTH: 25 MG
PACKING: 30 CAPSULES PER BOX
PRODUCT NAME: ISOFLURANE LIQUID FOR INHALATION
BRAND NAME: IFLURANE-100
STRENGTH: 100 ML
PACKING: SINGLE BOTTLE PER BOX
PRODUCT NAME: LIGNOCAINE HYDROCHLORIDE INJECTION
BRAND NAME:LIDFAST
STRENGTH: 2%, 30 ML
PACKING: 10 VIALS PER BOX

PRODUCT NAME:LIGNOCAINE HYDROCHLORIDE GEL (STERILE)
BRAND NAME:LIDFAST JELLY
STRENGTH: 2%, 30 GM
PACKING: SINGLE TUBE PER BOX
PRODUCT NAME: POTASSIUM CHLORIDE CONCENTRATE INJECTION
BRAND NAME: POTLYTE
STRENGTH: 15% W/V, 10 ML
PACKING: 5 AMPOULES PER BOX
PRODUCT NAME: PARACETAMOL INFUSION
BRAND NAME: PARAFUSE
STRENGTH: 10 MG / 1 ML, 100 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME: LABETALOL HCL INJECTION
BRAND NAME: LOBET
STRENGTH: 100 MG
PACKING: 50 TABLETS PER BOX
PRODUCT NAME: BIVALIRUDIN FOR INJECTION (LYOPHILIZED)
BRAND NAME: NGMINE
STRENGTH: 250 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME:CISATRACURIUM BESYLATE INJECTION
BRAND NAME: ATRACIS
STRENGTH: 10 MG / 5 ML
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DIGOXIN PEDIATRIC SYRUP
BRAND NAME: DIXIN
STRENGTH: 3 MG / 60 ML
PACKING: SINGLE BOTTLE PER BOX

PRODUCT NAME:DOBUTAMINE POWDER FOR INJECTION (LYOPHILIZED)
BRAND NAME: DOBUSTAT
STRENGTH: 250 MG
PACKING: SINGLE VIAL PER BOX

PRODUCT NAME: DOBUTAMINE HCL INJECTION
BRAND NAME: DOBUSTAT
STRENGTH: 250 MG / 5 ML
PACKING: 2 AMPOULES PER BOX

PRODUCT NAME:ENOXAPARIN SODIUM INJECTION (PREFILLED SYRINGE)
BRAND NAME: LOWCAP
STRENGTH: 40 MG / 0.4 ML
PACKING: SINGLE PFS PER BOX
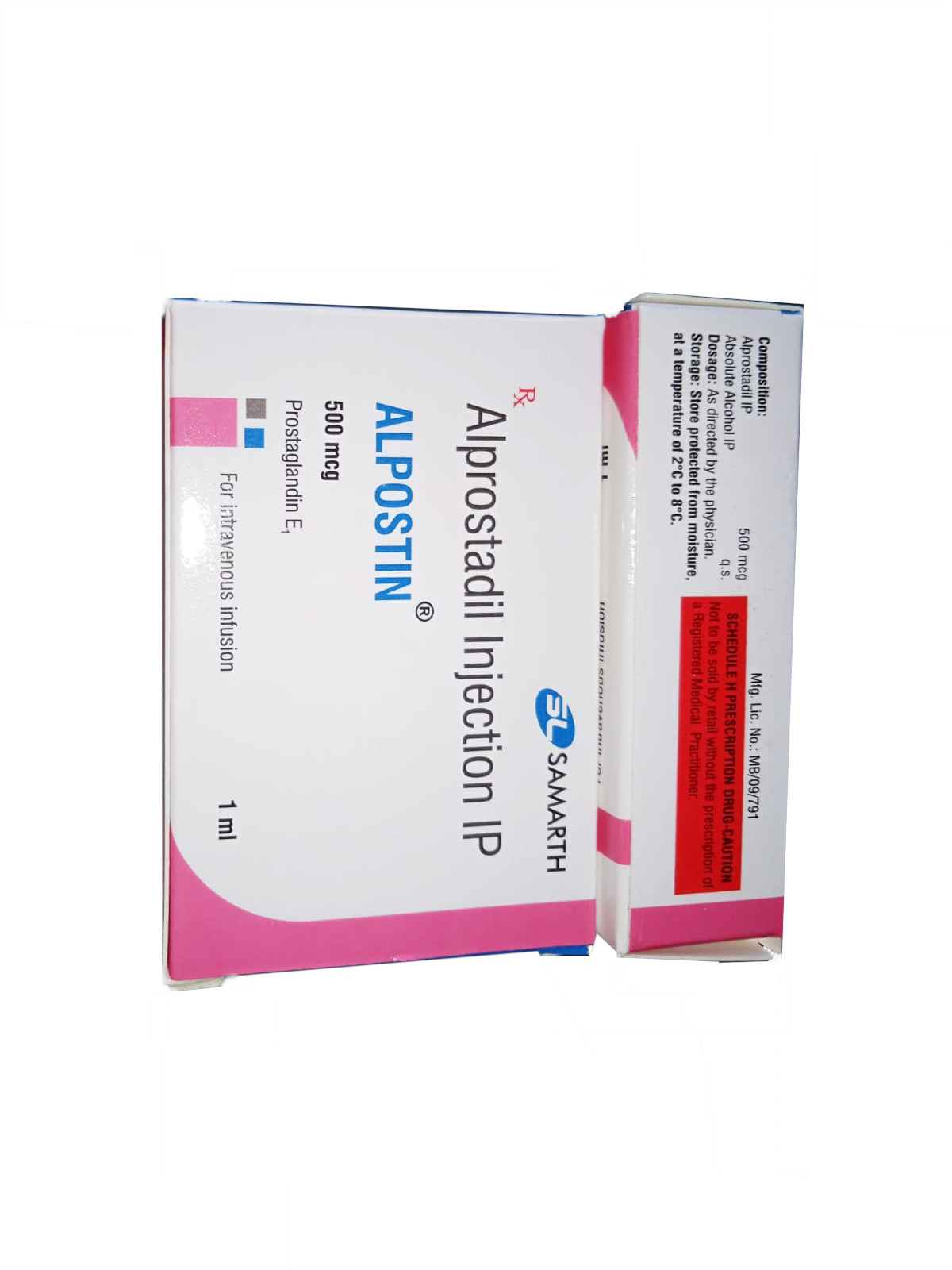
PRODUCT NAME: ALPROSTADIL (PROSTAGLANDIN E1) INJECTION
BRAND NAME: ALPOSTIN
STRENGTH: 500 MCG / 1 ML
PACKING: SINGLE AMPOULE PER BOX
WhatsApp us